Abstract
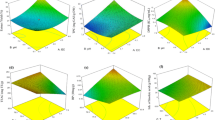
A maceration process was optimized using a full factorial design coupled to a response surface model for the extraction of phenolic compounds (PCs) from sugar maple leaves (SML). As part of the extraction process, high-speed homogenization (HSH) as a pre-treatment condition was investigated to promote the efficiency of maceration. Finally, characterization of SML was performed using colorimeter, SEM, FTIR spectroscopy, and LC-ESI-Q-TOF–MS/MS. The antioxidant activity of the extract was investigated as well using standard methods. Results showed that ethanol concentration and solid-to-solvent ratio had significant effects (p < 0.05) on the extraction of PCs and HSH can be considered as an effective pre-treatment towards maximizing phenolic compounds recovery from SML. Under the optimal conditions (ethanol concentration of 61.63% at a solid-to-solvent ratio of 1:38.22 g/mL), the experimental %total yield (28.57%), total phenolics (110.87 mg GAE/g DM), and total flavonoids (18.95 mg CTE/g DM) were in close agreement with the predicted values. A total of 81 PCs were tentatively identified including mostly phenolic acids and flavonoids and their derivatives from SML using LC-ESI-Q-TOF–MS/MS. What is more, a gallotannins pathway existing in SML from gallic acid to tetragalloyl-D-glucopyranose was proposed. The study suggested SML as a promising source of PCs which can be effectively extracted through maceration and be further used as natural antioxidants in the formulation of functional food systems.







Similar content being viewed by others
References
Kumar M, Dahuja A, Tiwari S, Punia S, Tak Y, Amarowicz R, Bhoite AG, Singh S, Joshi S, Panesar PS, Saini RP, Pihlanto A, Tomar M, Sharifi-Rad J, Kaur C (2021) Recent trends in extraction of plant bioactives using green technologies: A review. Food Chem 353:129431. https://doi.org/10.1016/j.foodchem.2021.129431
DiNardo A, Brar HS, Subramanian J, Singh A (2019) Optimization of microwave-assisted extraction parameters and characterization of phenolic compounds in Yellow European Plums. Can J Chem Eng 97(1):256–267
Martynenko A, Chen YG (2016) Degradation kinetics of total anthocyanins and formation of polymeric color in blueberry hydrothermodynamic (HTD) processing. J Food Eng 171:44–51
Sim YY, Ong WTE, Nyam KRL (2019) Effect of various solvents on the pulsed ultrasonic assisted extraction of phenolic compounds from Hibiscus cannabinus L. leaves. Industrial Crops and Products 140:8
Yeasmen N, Orsat V (2021) Green extraction and characterization of leaves phenolic compounds: a comprehensive review. Crit Rev Food Sci Nutr 1–39. https://doi.org/10.1080/10408398.2021.2013771
GrandViewResearch, Polyphenols market size, share & trends analysis report by product (grape seed, green tea, cocoa), by application (beverages, food, feed, dietary supplements, cosmetics), and segment forecasts, 2019–2025. 2019: California, United States. p. 154
Geoffroy TR, Fortin Y, Stevanovic T (2017) Hot-water extraction optimization of sugar maple (Acer saccharum Marsh.) and red maple (Acer rubrum L.) bark applying principal component analysis. Journal of Wood Chemistry and Technology 37(4):261–272
Honma A, Koyama T, Yazawa K (2010) Anti-hyperglycemic effects of sugar maple Acer saccharum and its constituent acertannin. Food Chem 123(2):390–394
Li L, Seeram NP (2010) Maple syrup phytochemicals include Lignans, Coumarins, a Stilbene, and other previously unreported antioxidant phenolic compounds. J Agric Food Chem 58(22):11673–11679
Li L, Seeram NP (2011) Further investigation into maple syrup yields 3 new lignans, a new phenylpropanoid, and 26 Other Phytochemicals. J Agric Food Chem 59(14):7708–7716
Yoshikawa K, Kawahara Y, Arihara S, Hashimoto T (2011) Aromatic compounds and their antioxidant activity of Acer saccharum. J Nat Med 65(1):191–193
Yoshikawa K, Tani S, Baba C, Hashimoto T (2013) Phenylpropanoid, sapnol A, lignan and neolignan sophorosides, saposides A and B, isolated from Canadian sugar maple sap. Molecules 18(8):9641–9649. https://doi.org/10.3390/molecules18089641
Yuan T, Wan C, González-Sarrías A, Kandhi V, Cech NB, Seeram NP (2011) Phenolic glycosides from sugar maple (Acer saccharum) bark. J Nat Prod 74(11):2472–2476
Contreras MD, Lama-Munoz A, Gutierrez-Perez JM, Espinola F, Moya M, Romero I, Castro E (2019) Integrated process for sequential extraction of bioactive phenolic compounds and proteins from mill and field olive leaves and effects on the lignocellulosic profile. Foods 8(11):17
Saini A, Panesar PS, Bera MB (2019) Valorization of fruits and vegetables waste through green extraction of bioactive compounds and their nanoemulsions-based delivery system. Bioresources and Bioprocessing 6. https://doi.org/10.1186/s40643-019-0261-9
Clodoveo ML, Crupi P, Annunziato A, Corbo F (2022) Innovative extraction technologies for development of functional ingredients based on polyphenols from olive leaves. Foods 11(1):103
FAOSTAT, Food and Agriculture Organization of the United Nations Database. 2020: Rome, Italy: FAO.
Sin K, Baraoidan WA, Gaspillo PAD (2014) Microwave-assisted extraction of phenolic compounds from Moringa oleifera Lam. Leaves using response surface methodology as optimization tool. Philippine Agricultural Scientist 97(1):36–42
Yeasmen N, Bhuiyan MHR, Orsat V (2021) Unravelling scientific research towards the green extraction of phenolic compounds from leaves: a bibliometric analysis. Int J Food Sci Technol 56(10):4893–4906
Kaur GJ, Orsat V, Singh A (2022) Sustainable usage of carrot discards in food processing. Int J Sust Dev World 29(1):18–26
Akhlisah ZN, Yunus R, Abidin ZZ, Lim BY, Kania D (2021) Pretreatment methods for an effective conversion of oil palm biomass into sugars and high-value chemicals. Biomass Bioenerg 144:105901
Alanon ME, Ivanovic M, Gomez-Caravaca AM, Arraez-Roman D, Segura-Carretero A (2020) Choline chloride derivative-based deep eutectic liquids as novel green alternative solvents for extraction of phenolic compounds from olive leaf. Arab J Chem 13(1):1685–1701
Cujic N, Savikin K, Jankovic T, Pljevljakusic D, Zdunic G, Ibric S (2016) Optimization of polyphenols extraction from dried chokeberry using maceration as traditional technique. Food Chem 194:135–142
Sandra P, Zoric Z, Kovacevic DB, Garofulic IE, Dragovic-Uzelac V (2020) Pressurized hot water extraction of phenolic compounds from leaves of Stevia rebaudiana: An UPLC-ESI-MSMS study. J Food Process Eng 43(2):10
Uysal S, Cvetanovic A, Zengin G, Zekovic Z, Mahomoodally MF, Bera O (2019) Optimization of maceration conditions for improving the extraction of phenolic compounds and antioxidant effects of Momordica Charantia L. leaves through response surface methodology (RSM) and artificial neural networks (ANNs). Analytical Letters 52(13):2150–2163
Hannachi H, Benmoussa H, Saadaoui E, Saanoun I, Negri N, Elfalleh W (2019) Optimization of ultrasound and microwave-assisted extraction of phenolic compounds from olive leaves by response surface methodology. Research Journal of Biotechnology 14(7):28–37
Garmus TT, Paviani LC, Queiroga CL, Cabral FA (2015) Extraction of phenolic compounds from pepper-rosmarin (Lippia sidoides Cham.) leaves by sequential extraction in fixed bed extractor using supercritical CO2, ethanol and water as solvents. Journal of Supercritical Fluids 99:68–75
Jiao J, Gai QY, Zhang L, Wang W, Luo M, Zu YG, Fu YJ (2015) High-speed homogenization coupled with microwave-assisted extraction followed by liquid chromatography-tandem mass spectrometry for the direct determination of alkaloids and flavonoids in fresh Isatis tinctoria L. hairy root cultures. Analytical and Bioanalytical Chemistry 407(16):4841–4848
Pei WJ, Guo RL, Zhang JL, Li XQ (2019) Extraction of phenylethanoid glycosides from Cistanche tubulosa by high-speed shearing homogenization extraction. J AOAC Int 102(1):63–68
UNSDGs (2015) 2030 Agenda for Sustainable Development [Online]. New York city, USA: UNITED NATIONS. Available: https://www.un.org/sustainabledevelopment/development-agenda. Accessed 25 Oct 2021
Singleton VL, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. In: Oxidants and antioxidants, Pt A, Packer L (ed) 299:152–178
Jia Z, Tang MC, Wu JM (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 64(4):555–559
Thaipong K, Boonprakob U, Crosby K, Cisneros-Zevallos L, Byrne DH (2006) Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J Food Compos Anal 19(6–7):669–675
Benzie IFF, Strain JJ (1999) Ferric reducing antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration, in Oxidants and Antioxidants, Pt A, Packer L (ed) 299:15–27
Rafiee Z, Jafari SM, Alami M, Khomeiri M (2011) Microwave-assisted extraction of phenolic compounds from olive leaves; a comparison with maceration. Journal of Animal and Plant Sciences 21(4):738–745
HealthCanada (2015) List of permitted carrier or extraction solvents (lists of permitted food additives) [Online]. Canada: Government of Canada. Available: https://www.canada.ca/en/health-canada/services/food-nutrition/food-safety/food-additives/lists-permitted/15-carrier-extraction-solvents-2016-06-28.html. Accessed 20 Jan 2022
USFDA (2019) Food additive status list [Online]. United States. Available: https://www.fda.gov/food/food-additives-petitions/food-additive-status-list#ftnE. Accessed 20 Jan 2022
da Rosa GS, Vanga SK, Gariepy Y, Raghavan V (2019) Comparison of microwave, ultrasonic and conventional techniques for extraction of bioactive compounds from olive leaves (Olea europaea L.). Innovative Food Sci Emerg Technol 58. https://doi.org/10.1016/j.ifset.2019.102234
Elboughdiri N (2018) Effect of time, solvent-solid ratio, ethanol concentration and temperature on extraction yield of phenolic compounds from olive leaves. Engineering Technology & Applied Science Research 8(2):2805–2808
Krakowska A, Rafinska K, Walczak J, Kowalkowski T, Buszewski B (2017) Comparison of various extraction techniques of Medicago sativa: yield, antioxidant activity, and content of phytochemical constituents. J AOAC Int 100(6):1681–1693
Zhao BB, Deng JW, Li H, He YQ, Lan T, Wu D, Gong HD, Zhang Y, Chen ZC (2019) Optimization of phenolic compound extraction from Chinese Moringa oleifera leaves and antioxidant activities. J Food Qual 2019:13
Crupi P, Dipalmo T, Clodoveo ML, Toci AT, Coletta A (2018) Seedless table grape residues as a source of polyphenols: comparison and optimization of non-conventional extraction techniques. Eur Food Res Technol 244(6):1091–1100
Xu SH, Wang GY, Guo RL, Wei Z, Zhang JL (2019) Extraction of steviol glycosides from Stevia rebaudiana (Bertoni) leaves by high-speed shear homogenization extraction. J Food Process Preserv 43(12). https://doi.org/10.1111/jfpp.14250
Ozgen M, Reese RN, Tulio AZ Jr, Scheerens JC, Miller AR (2006) Modified 2,2-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) method to measure antioxidant capacity of selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2′-diphenyl-1- picrylhydrazyl (DPPH) methods. J Agric Food Chem 54(4):1151–1157
Deng Y, Yang GY, Yue J, Qian BJ, Liu ZM, Wang DF, Zhong Y, Zhao YY (2014) Influences of ripening stages and extracting solvents on the polyphenolic compounds, antimicrobial and antioxidant activities of blueberry leaf extracts. Food Control 38:184–191
Nongmaithema R, Meda V (2017) Optimization of microwave vacuum drying parameters for germinated lentils based on starch digestibility, antioxidant activity and total phenolic content. International Journal of Food Studies 6:44–55
Rahimi J, Ngadi MO (2016) Structure and irregularities of surface of fried batters studied by fractal dimension and lacunarity analysis. Food Structure-Netherlands 9:13–21
Sansiribhan S, Devahastin S, Soponronnarit S (2012) Generalized microstructural change and structure-quality indicators of a food product undergoing different drying methods and conditions. J Food Eng 109(1):148–154
Alara OR, Abdurahman NH, Ukaegbu CI (2018) Soxhlet extraction of phenolic compounds from Vernonia cinerea leaves and its antioxidant activity. Journal of Applied Research on Medicinal and Aromatic Plants 11:12–17
Wishart DS, Feunang YD, Marcu A, Guo AC, Liang K, Vazquez-Fresno R, Sajed T, Johnson D, Li CR, Karu N, Sayeeda Z, Lo E, Assempour N, Berjanskii M, Singhal S, Arndt D, Liang YJ, Badran H, Grant J, Serra-Cayuela A, Liu YF, Mandal R, Neveu V, Pon A, Knox C, Wilson M, Manach C, Scalbert A (2018) HMDB 4.0: the human metabolome database for 2018. Nucleic Acids Research 46(D1):D608–D617
Rothwell JA, Perez-Jimenez J, Neveu V, Medina-Remon A, M’Hiri N, Garcia-Lobato P, Manach C, Knox C, Eisner R, Wishart DS, Scalbert A (2013) Phenol-Explorer 3.0: a major update of the Phenol-Explorer database to incorporate data on the effects of food processing on polyphenol content. Database-the Journal of Biological Databases and Curation. https://doi.org/10.1093/database/bat070
Belwal T, Pandey A, Bhatt ID, Rawal RS, Luo ZS (2019) Trends of polyphenolics and anthocyanins accumulation along ripening stages of wild edible fruits of Indian Himalayan region. Sci Rep 9. https://doi.org/10.1038/s41598-019-42270-2
Bhuyan DJ, Basu A (2017) Phenolic compounds potential health benefits and toxicity. Utilisation of Bioactive Compounds from Agricultural and Food Production Waste. CRC Press, pp 27–59
Cory H, Passarelli S, Szeto J, Tamez M, Mattei J (2018) The role of polyphenols in human health and food systems: a mini-review. Front Nutr 5. https://doi.org/10.3389/fnut.2018.00087
Kumar N, Goel N (2019) Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnology Reports 24. https://doi.org/10.1016/j.btre.2019.e00370
Saltveit ME (2017) Synthesis and metabolism of phenolic compounds, in Fruit and vegetable phytochemicals: chemistry and human health: second edition. Wiley Blackwell. p. 115–123
Yang LG, Yin PP, Fan H, Xue Q, Li K, Li X, Sun LW, Liu YJ (2017) Response surface methodology optimization of ultrasonic-assisted extraction of Acer Truncatum leaves for maximal phenolic yield and antioxidant activity. Molecules 22(2):21
Acknowledgements
The authors are also thankful to Professor Saji George, Department of Food Science and Agricultural Chemistry, McGill University, for allowing the use of plate reader (SpectraMax® i3x, Multi-mode microplate reader, Molecular Devices, USA) at no cost.
Funding
The authors received funding support from IDB-McGill Scholarship and Natural Science and Engineering Research Council (NSERC), Canada.
Author information
Authors and Affiliations
Contributions
Nushrat Yeasmen — conceptualization; data curation; formal analysis; methodology; writing — original draft, review and editing. Valérie Orsat — supervision; writing — review and editing.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yeasmen, N., Orsat, V. Maximization of the recovery of phenolic compounds from sugar maple leaves. Biomass Conv. Bioref. 14, 6251–6266 (2024). https://doi.org/10.1007/s13399-022-02904-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-02904-4