Abstract
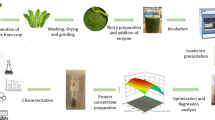
Radish leaf protein concentrates (RLPC) were prepared by alkaline extraction and characterized for their antioxidant activity, functional properties, mineral content, in-vitro digestibility and microbial stability. Numerical optimization using the 3-factor Box–Behnken Design of response surface methodology suggested that the optimized extraction was obtained at a pH of 9.46, sample/water ratio of 0.075 and time of extraction 46.89 min resulting in 12.12% yield of RLPC with protein content of 87.64%. Glutelins (41.49%), prolamins (24.96%) and albumins (20.43%) were found to be the three major fractions of the protein concentrate, while globulins (13.00%) contributed as a minor component. The apparent molecular weights of these protein fractions ranged between 14 and 60 kDa. Antioxidant activities (FRAP, ABTS and DPPH) were higher in RLPC as compared to the isolated fractions. Functional properties like water holding capacity, oil holding capacity, emulsifying capacity and emulsion stability of the RLPC were 352, 280, 48.1 and 47.8%, respectively. Ca and Fe were the most abundant major and trace minerals, respectively, present in the RLPC. In-vitro protein digestibility was found to be 93.51% and its microbial load remained in acceptable limits during 42 days of storage under both refrigerated and ambient temperature conditions. Our results indicated that the protein concentrates extracted from radish leaves have considerable antioxidant activity, functional properties, mineral content, digestibility and microbial stability. The results highlight the potential of RLPC for use in functional foods as a safe and cost-effective source of protein.






Similar content being viewed by others
References
A. Rani, Y. Arfat, R.S. Aziz, L. Ali, H. Ahmed, S. Asim, M. Rashid, C.H. Hocart, Environ. Technol. Innov. 23, 1–13 (2021). https://doi.org/10.1016/j.eti.2021.101620
Ankita, K. Prasad, Pharm. Lett. 7, 269–279 (2015)
K.V. Badar, A.U. Kulkarni, Curr. Bot. 2, 5–7 (2011)
A.E. Ghaly, F.N. Alkoaik, Am. J. Appl. Sci. (2010). https://doi.org/10.3844/ajassp.2010.331.342
S. Jiamyangyuen, V. Srijesdaruk, W.J. Harper, Extension 27, 56 (2005)
S. Tang, N.S. Hettiararchy, S. Eswaranandam, P. Crandall, Food Sci. (2003). https://doi.org/10.1111/j.1365-2621.2003.tb05696.x
A.O. Fasuyi, V.A. Aletor, Pak. J. Nutr. (2005). https://doi.org/10.3923/pjn.2005.43.49
P.F. Coldebella, S.D. Gomes, J.A. Evarini, M.P. Cereda, S.R. Coelho, A. Coldebella, Eng. Agric. 33, 1223–1233 (2013)
M.H. Soo, N.A. Samad, D.N.A. Zaidel, Y.M.M. Jusoh, I.I. Muhamad, Z. Hashim, Chem. Eng. Trans. (2021). https://doi.org/10.3303/CET2189043
S. Rawdkuen, Food Appl. Biosci. J. 8, 43–67 (2020)
M. Contreras, A. Lama-Muñoz, J.G.P. Manuel, F. Espínola, M. Moya, E. Castro, Bioresour Technol. (2019). https://doi.org/10.1016/j.biortech.2019.02.040
C. Zhang, J.P.M. Sanders, T.T. Xiao, M.E. Burns, PLoS ONE (2015). https://doi.org/10.1371/journal.pone.0133046
S.O. Ogunwolu, F.O. Henshaw, H.P. Mock, A. Santros, S.O. Awonorin, Food Chem. (2009). https://doi.org/10.1016/j.foodchem.2009.01.011
A.A. Wani, D.S. Sogi, L. Grover, D.C. Saxena, Biosyst. Eng. (2006). https://doi.org/10.1016/j.biosystemseng.2006.02.004
AOAC, Official Methods of Analysis, 17th edn. (Association of Official Analytical Chemists, Washington, DC, 2000)
A.P. Adebiyi, R.E. Aluko, Food Chem. (2011). https://doi.org/10.1016/j.foodchem.2011.03.116
O.H. Lowry, N.J. Rosebrough, A.L. Farr, R.J. Randall, J. Biol. Chem. 193, 265 (1951)
U.K. Laemmli, Nature 227, 680–685 (1970)
C.W. Lin, C.W. Yu, K.H. Yih, J. Food Drug Anal. 5, 386–395 (2009)
R. Re, N. Pellegrini, A. Proteggente, A. Pannala, M. Yang, C. Rice-Evans, Free Radic. Biol. Med. (1999). https://doi.org/10.1016/s0891-5849(98)00315-3
I.F. Benzie, J.J. Strain, Anal. Biochem. (1996). https://doi.org/10.1006/abio.1996.0292
W.E. Hillis, T. Swain, J. Sci. Food Agric. 10, 135–144 (1969). https://doi.org/10.1002/jsfa.2740100211
I.S. Balbaa, A.Y. Zaki, A.M. El Shamy, J. Assoc. Off. Anal. Chem. 57, 752–755 (1974). https://doi.org/10.1093/jaoac/57.3.752
J. Yu, M. Ahmedna, I. Goktepe, Food Chem. (2007). https://doi.org/10.1016/j.foodchem.2006.08.012
M.J.Y. Lin, E.S. Humbert, F.W. Sosulski, J. Food Sci. (1974). https://doi.org/10.1111/j.1365-2621.1974.tb02896.x
K. Yasumatsu, K. Sawada, S. Moritaka, M. Misaki, J. Toda, T. Wada, K. Ishii, Agric. Biol. Chem. (1972). https://doi.org/10.1080/00021369.1972.108603212
C.W. Coffman, V.V. Garcia, J. Food Tech. 12, 473–484 (1977)
N. Huda, A. Abdullah, A.S. Babji, Int. J. Food Sci. Technol. (2001). https://doi.org/10.1046/j.1365-2621.2001.00473.x
J.F. Pedler, D.R. Parker, D.E. Crowley, Planta (2000). https://doi.org/10.1007/s004250000270
D. Znidarcic, T. Pozrl, Acta. Agric. Slov. 87, 235–243 (2006)
M. Minekus, M. Alminger, P. Alvito, S. Balance, Brodkorb A. Food Funct. (2014). https://doi.org/10.1039/C3FO60702J
V. Nour, I. Trandafir, S. Cosmulescu, J. Chrom. Sci. 51, 883–890 (2013). https://doi.org/10.1093/chromsci/bms180
R. Aslam, M.S. Alam, S. Singh, S. Kumar, LWT 151, 112183 (2021). https://doi.org/10.1016/j.lwt.2021.112183
G. Kaur, S. Bhatia, Int. J. Agric. Sci. (2021). https://doi.org/10.15740/HAS/IJAS/17.2/185-193
G.S. Mann, S. Bhatia, M.S. Alam, Agric. Eng. Int. CIGRJ. 4, 243–251 (2016)
A. Akyuz, S. Ersus, Food Chem. 335, 127673 (2021). https://doi.org/10.1016/j.foodchem.2020.127673
M. Wang, N.S. Hettiarachchy, M. Qi, W. Burks, T. Siebenmorgen, J Agric. Food Chem. (1999). https://doi.org/10.1021/jf9806964
I.S. Rustom, M.H. Lopex-Leiva, B.M. Nair, Food Sci. (1991). https://doi.org/10.1016/0308-8146(91)90031-I
D.N. López, M. Galante, M. Robson, V. Boeris, D. Spelzini, Int. J. Biol. Macromol. (2018). https://doi.org/10.1016/j.ijbiomac.2017.12.080
M. Çelik, M. Güzel, M. Yildirim, J. Food Sci. Technol. (2019). https://doi.org/10.1007/s13197-019-03785-8
B.P. Lamsal, R.G. Koegel, S. Gunasekaran, LWT Food Sci. Technol. (2007). https://doi.org/10.1016/j.lwt.2006.11.010
A.A. Famuwagun, A.M. Alashi, S.O. Gbadamosi, K.A. Taiwo, D.J. Oyedele, O.C. Adebooye, R.E. Aluko, Int. J. Food Prop. (2020). https://doi.org/10.1080/10942912.2020.1772285
A.P. Adebiyi, A.O. Adebiyi, Y. Hasegawa, T. Ogawa, K. Muramoto, Eur. Food Res. Technol. (2009). https://doi.org/10.1007/s00217-008-0945-4
J. Dai, R.J. Mumper, Molecules (2010). https://doi.org/10.3390/molecules15107313
O. Kadiri, C.T. Akanbi, B.T. Olawoye, S.O. Gbadamosi, Int. J. Food Prop. (2017). https://doi.org/10.1080/10942912.2016.1230874
Z. Xie, J. Huang, X. Xu, Z. Jin, Food Chem. (2008). https://doi.org/10.1016/j.foodchem.2008.03.078
M. Delfanian, S.M. Razavi, M.H.H. Khodaparast, R.E. Kenari, S. Golmohammadzadeh, Food Res. Int. (2018). https://doi.org/10.1016/j.foodres.2018.03.043
A. Sodamade, S.M. Raimi, A.D. Owonikoko, A.T. Adebimpe, IJTSRD. 36, 57–68 (2019)
A. Fernández-Quintela, M.T. Macarulla, A.S. Del Barrio, J.A. Martínez, Plant Food Hum. Nutr. 51, 331–341 (1997)
P.C. Sharma, B.M.K.S. Tilakratne, A. Gupta, J. Food Sci. Technol. (2010). https://doi.org/10.1007/s13197-010-0096-z
S.K. Sathe, S.S. Deshpande, D.K. Salunkhe, J. Food Sci. (1982). https://doi.org/10.1111/j.1365-2621.1982.tb10110.x
S. Damodaran, Food Sci. Technol. 1, 1–24 (1997)
A. Subagio, Food Chem. (2006). https://doi.org/10.1016/j.foodchem.2004.12.042
A.M. Ghribi, I.M. Gafsi, C. Blecker, S. Danthine, H. Attia, S. Besbes, J. Food Eng. (2015). https://doi.org/10.1016/j.jfoodeng.2015.06.021
O. Aletor, A. Oshodi, K. Ipinmoroti, Food Chem. (2002). https://doi.org/10.1016/S0308-8146(01)00376-4
R. Chatterjee, T.K. Dey, M. Ghosh, P. Dhar, Food Bioprod. Process 94, 70–81 (2015)
M. Du, J. Xie, B. Gong, X. Xu, W. Tang, X. Li, C. Li, M. Xie, Food Hydrocoll. (2018). https://doi.org/10.1016/j.foodhyd.2017.01.003
K. Shevkani, N. Singh, A. Kaur, J.C. Rana, Food Hydrocoll. 43, 679–689 (2015)
J. Cheng, S. Zhou, D. Wu, J. Chen, D. Liu, X. Ye, Food Chem. (2009). https://doi.org/10.1016/j.foodchem.2008.05.106
F. Garcia-Moreno, E. Solórzano, J. Banhart, Soft Matter 7, 9216–9223 (2011)
H. Wu, Q. Wang, T. Ma, J. Ren, Food Res. Int. (2009). https://doi.org/10.1016/j.foodres.2008.12.006
T.G. Kudre, S. Benjakul, H. Kishimura, J. Sci. Food Agric. (2013). https://doi.org/10.1002/jsfa.6052
NRC, National Research Council (1989) https://doi.org/10.17226/1349
S.A. El Sohaimy, G.M. Hamad, S.E. Mohamed, M.H. Amar, R.R. Al-Hindi, Global Adv. Res. J. Agric. Sci. 4, 188–199 (2015)
J.O. Agbede, J. Sci. Food Agr. (2006). https://doi.org/10.1002/jsfa.2491
J.K. Mensah, R.I. Okoli, J.O. Ohaju-Obodo, K. Eifediyi, Afr. J. Biotechnol. 7, 14 (2008)
L.H. Khan, V.K. Varshney, J. Diet Suppl. (2018). https://doi.org/10.1080/19390211.2017.1349232
R. Toews, N. Wang, Food Res. Int. (2013). https://doi.org/10.1016/j.foodres.2012.12.009
G. Pumilia, M.J. Cichon, J.L. Cooperstone, G. Dugo, S.J. Schwartz, Food Res. Int. (2014). https://doi.org/10.1016/j.foodres.2014.05.047
J.Y. Han, K. Khan, Cereal Chem. 67, 384–390 (1990)
Y.A. Adebowale, I.A. Adeyemi, A.A. Oshodi, K. Niranjan, Food Chem. (2007). https://doi.org/10.1016/j.foodchem.2006.11.05
T.A. Aderinola, A.M. Alashi, I.D. Nwachukwu, T.N. Fagbemi, T.N. Enujiugha, R.E. Aluko, Food Hydrocoll. (2020). https://doi.org/10.1016/j.foodhyd.2019.105574
T. Benhammouche, A. Melo, Z. Martins, M.A. Faria, S.C. Pinho, I.M. Ferreira, F. Zaidi, Food Chem. (2021). https://doi.org/10.1016/j.foodchem.2020.128858
M. Corgneau, C. Gaiani, J. Petit, Y. Nikolova, S. Banon, L. Riti’e-Pertusa, D.T.L. Le, J. Scher, Int. J. Dood Sci. Technol. (2019). https://doi.org/10.1111/ijfs.14170
P. Goufo, I. Cortez, Biology (2020). https://doi.org/10.3390/biology9090268
R. Flamini, Mass Spec. Rev. 22, 218–250 (2008). https://doi.org/10.1002/mas.10052
FSSAI Specification (2018) for dehydrated vegetable products. https://archive.fssai.gov.in
R. Sasikumar, K. Vivek, A.K. Jaiswal, J. Food Process. Preserv. (2021). https://doi.org/10.1111/jfpp.15568
Acknowledgements
Authors are grateful to Dr Tarsem Singh Dhillon, Associate Director (Seeds), Punjab Agricultural University, Ludhiana for providing radish leaves and Department of Processing and Food Engineering, Punjab Agricultural University, Ludhiana for providing facilities for carrying out the research experiments. The authors are also thankful to Dr Amrit Kaur Mahal (Professor of Statistics) for her guidance in statistical analysis during research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kaur, G., Bhatia, S. Radish leaf protein concentrates: optimization of alkaline extraction for production and characterization of an alternative plant protein concentrate. Food Measure 16, 3166–3181 (2022). https://doi.org/10.1007/s11694-022-01411-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-022-01411-4