Abstract
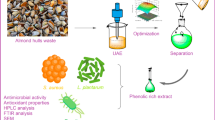
In this study, antioxidant (DPPH and metal chelating), DNA cleavage, biofilm, and antimicrobial properties of extracted phenol from the walnut green husk (WGH) and its different concentrate and permeate samples were evaluated. For maximum phenolic compound extraction from the WGH first, the effects of solvent type (deionized water, methanol, n-hexane, acetone, and ethanol), solvent temperature (25–75 °C), and extraction time (0.5–24 h) were optimized. Then to concentrate phenolic compounds a pressure-driven membrane process was used with four different membrane types. The phenol contents of the concentrate samples were found to be microfiltration (MF) concentrate 4400 mg/L, ultrafiltration (UF) concentrate 4175 mg/L, nanofiltration (NF) concentrate 8155 mg/L, and reverse osmosis (RO) concentrate 8100 mg/L. LC-MSMS was used to determine the quantification of phenolic compounds in permeate and concentrate streams. In addition, all of the concentrate samples with high phenol content showed a high antioxidant activity as 100% with MF concentrate, UF concentrate, NF concentrated and RO concentrated. Likewise, concentrate samples were found to have very high antibiofilm activity as 82.86% for NF concentrate againts S. aureus, 85.80% for NF concentrate against P. aureginosa, 80.95% for RO concentrate against S. aureus, and 83.61% for RO-concentrate against P. aureginosa. When the antimicrobial activity of the extracted phenol from WGH and its different concentrate and permeate samples were evaluated by micro dilution and disk diffusion methods, it was found that the ability of the concentrate samples to inhibit bacterial growth was much higher than permeate ones. In addition, extracted phenol from WGH and its different concentrate and permeate samples showed significant DNA nuclease activity.





Similar content being viewed by others
Data availability statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- DPPH:
-
2, 2-Diphenyl-1-picrylhydrazyl
- MF:
-
Microfiltration
- MIC:
-
Minimum inhibition concentration
- NF:
-
Nanofiltration
- RO:
-
Reverse osmosis
- UF:
-
Ultrafiltration
- WGH:
-
Walnut green husk
References
Ağırtaş M, Karataş C, Ozdemir S (2015) Synthesis of some metallophthalocyanineswith dimethyl 5-(phenoxy)-isophthalate substituents and evaluation of theirantioxidant-antibacterial activities. Spectrochim Acta Part A Mol Biomolspectrosc 135:20–24
Agullo A, Pereira E, Freire M, Valentão P, Andrade P, Alvarez J et al (2013) Influence of solvent on the antioxidant and antimicrobial properties of walnut (Juglans regia L.) green husk extracts. Ind Crops Products 42:126–132
Akinmoladun AC, Falaiye E, Ojo OB, Adeoti A, Amoo ZA, Olaleye MT (2022) Effect of extraction technique, solvent polarity, and plant matrix on the antioxidant properties of Chrysophyllum albidum G Don. (African Star Apple). Bull Natl Res Centre. https://doi.org/10.1186/s42269-022-00718-y
Aminov R (2010) A brief history of the antibiotic era: lessons learned and challenges for the future. Front Microbiol 8(1):134. https://doi.org/10.3389/fmicb.2010.00134
Arend GD, Adorno WT, Rezzadori K, Di Luccio M, Chaves VC, Reginatto FH, Petrus JCC (2017) Concentration of phenolic compounds from strawberry (Fragaria X ananassa Duch) juice by nanofiltration membrane. J Food Eng 201:36–41. https://doi.org/10.1016/J.JFOODENG.2017.01.014
Caballero E, Soto C, Jara J (2019) Thermal stability data of juglone from extracts of walnut (Juglans regia) green husk, and technologies used to concentrate juglone. Data Brief 25:104081. https://doi.org/10.1016/J.DIB.2019.104081
Carvalho M, Ferreira PJ, Mendes VS, Silva R, Pereira JA, Jerónimo C, Silva BM (2010) Human cancer cell antiproliferative and antioxidant activities of Juglans regia L. Food Chem Toxicol 48(1):441–447. https://doi.org/10.1016/J.FCT.2009.10.043
Chandramohan Y, Padmanaban V, Bethunaickan R, Tripathy S, Swaminathan S, Ranganathan UD (2019) In vitro interaction profiles of the new antitubercular drugs bedaquiline and delamanid with moxifloxacin against clinical Mycobacterium tuberculosis isolates. J Glob Antimicrob Resist 19:348–353. https://doi.org/10.1016/j.jgar.2019.06.013
Che Sulaiman IS, Basri M, Fard Masoumi HR, Chee WJ, Ashari SE, Ismail M (2017) Effects of temperature, time, and solvent ratio on the extraction of phenolic compounds and the anti-radical activity of Clinacanthus nutans Lindau leaves by response surface methodology. Chem Cent J 11(1):1–11. https://doi.org/10.1186/s13065-017-0285-1
Darvishi E, Kahrizi D, Arkan E (2019) Comparison of different properties of zinc oxide nanoparticles synthesized by the green (using Juglans regia L. leaf extract) and chemical methods. J Mol Liq 286:110831. https://doi.org/10.1016/j.molliq.2019.04.108
Dolatabadi S, Moghadam HN, Mahdavi-Ourtakand M (2018) Evaluating the anti -biofilm and antibacterial effects of Juglans regia L. extracts against clinical isolates of Pseudomonas aeruginosa. Microb Pathog 118:285–289. https://doi.org/10.1016/j.micpath.2018.03.055
FAO (2020) Statistical Yearbook. Agricultural production. https://www.fao.org/home/en
Fernández-Agulló A, Pereira E, Freire MS, Valentão P, Andrade PB, González-álvarez J, Pereira JA (2013) Influence of solvent on the antioxidant and antimicrobial properties of walnut (Juglans regia L.) green husk extracts. Ind Crops Products 42(1):126–132. https://doi.org/10.1016/J.INDCROP.2012.05.021
Goli AH, Barzegar M, Sahari MA (2005) Antioxidant activity and total phenolic compounds of pistachio (Pistachia vera) hull extracts. Food Chem 92(3):521–525. https://doi.org/10.1016/j.foodchem.2004.08.020
Gomes F, Martins N, Barros L, Rodrigues ME, Oliveira M, Beatriz PP, Henriques M, Ferreira ICFR (2018) Plant phenolic extracts as an effective strategy to control Staphylococcus aureus the dairy industry pathogen. Ind Crops Prod 112:515–520. https://doi.org/10.1016/j.indcrop.2017.12.027
Jahanban-Esfahlan A, Jahanban-Esfahlan R, Tabibiazar M, Roufegarinejad L, Amarowicz R (2020) Recent advances in the use of walnut (Juglans regia L.) shell as a valuable plant-based bio-sorbent for the removal of hazardous materials. Res Adv 10:7026. https://doi.org/10.1039/c9ra10084a
Jaitz L, Siegl K, Eder R, Rak G, Abranko L, Koellensperger G, Hann S (2010) LC–MS/MS analysis of phenols for classification of red wine according to geographic origin grape variety and vintage. Food Chem 122(1):366–372. https://doi.org/10.1016/j.foodchem.2010.02.053
Kadiroğlu P, Ekici H (2018) Yeşil ceviz kabuklarının biyoaktif özelliklerinin FT-IR spektroskopi yöntemiyle tahmin edilmesi. Akademik Gıda 16(1):20–26. https://doi.org/10.24323/AKADEMIK-GIDA.415643
Karimi N, Minaei S, Hasani D, Eyvani A (2008) Parameters involved in walnut peeling process. J Agric Machinery Sci 4(4):389–393
Kavak DD, Altıok E, Bayraktar O, Ülkü S (2010) Pistacia terebinthus extract: As a potential antioxidant antimicrobial and possible β-glucuronidase inhibitor. J Mol Catal B Enzym 64(3–4):167–171. https://doi.org/10.1016/j.molcatb.2010.01.029
Kong X, Bao S, Song W, Hua Y, Zhang C, Chen Y, Li X (2021) Contributions of ethanol fractionation on the properties of vegetable protein hydrolysates and differences in the characteristics of metal (Ca Zn Fe)-chelating peptides. LWT 146:111482. https://doi.org/10.1016/j.lwt.2021.111482
Lee JH, Kim YG, Khadke SK, Yamano A, Woo JT, Lee J (2019) Antimicrobial and antibiofilm activities of prenylated flavanones from Macaranga tanarius. Phytomedicine 63:153033. https://doi.org/10.1016/j.phymed.2019.153033
Li X, Deng S (2020) Synergistic inhibition effect of walnut green husk extract and potassium iodide on the corrosion of cold rolled steel in trichloroacetic acid solution. J Market Res 9(6):15604–15620. https://doi.org/10.1016/J.JMRT.2020.11.018
Lu C, Liu H, Shangguan W, Chen S, Zhong Q (2021) Antibiofilm activities of the cinnamon extract against Vibrio parahaemolyticus and Escherichia coli. Arch Microbiol 203(1):125–135. https://doi.org/10.1007/s00203-020-02008-5
Lv Y, Wei K, Meng X, Huang Y, Zhang T, Li Z (2017) Separation and identification of iron-chelating peptides from defatted walnut flake by nanoLC-ESI–MS/MS and de novo sequencing. Process Biochem 59:223–228. https://doi.org/10.1016/j.procbio.2017.05.010
Machado MTC, Mello BCBS, Hubinger MD (2015) Evaluation of pequi (Caryocar Brasiliense Camb.) aqueous extract quality processed by membranes. Food Bioprod Process 95:304–312. https://doi.org/10.1016/J.FBP.2014.10.013
Mehdizadeh T, Zamani A, Froushani SMA (2020) Preparation of Cu nanoparticles fixed on cellulosic walnut shell material and investigation of its antibacterial antioxidant and anticancer effects. Heliyon 6(3):e03528. https://doi.org/10.1016/j.heliyon.2020.e03528
Mokrani A, Madani K (2016) Effect of solvent, time and temperature on the extraction of phenolic compounds and antioxidant capacity of peach (Prunus persica L.) fruit. Sep Purif Technol 162:68–76. https://doi.org/10.1016/J.SEPPUR.2016.01.043
Naczk M, Shaidi F (2004) Extraction and analysis of phenolics in food. J Chromatogr A 1504:95–111
Nile SH, Nile A, Liu J, Kim DH, Kai G (2019) Exploitation of apple pomace towards extraction of triterpenic acids antioxidant potential cytotoxic effects and inhibition of clinically important enzymes. Food Chem Toxicol 131:110563. https://doi.org/10.1016/j.fct.2019.110563
Oliveira I, Sousa A, Ferreira ICFR, Bento A, Estevinho L, Pereira JA (2008a) Total phenols, antioxidant potential and antimicrobial activity of walnut (Juglans regia L.) green husks. Food Chem Toxicol 46(7):2326–2331. https://doi.org/10.1016/J.FCT.2008.03.017
Ozay Y, Ozdemir S, Gonca S, Canli O, Dizge N (2021) Phenolic compounds recovery from pistachio hull using pressure-driven membrane process and a cleaner production of biopesticide. Environ Technol Innov 24:101993. https://doi.org/10.1016/J.ETI.2021.101993
Pinelo M, Rubilar M, Jerez M, Sineiro J, Núñez MJ (2005) Effect of solvent, temperature, and solvent-to-solid ratio on the total phenolic content and antiradical activity of extracts from different components of grape pomace. J Agric Food Chem 53(6):2111–2117. https://doi.org/10.1021/jf0488110
Ramezani N, Raji F, Rezakazemi M, Younas M (2020) Juglone extraction from walnut (Juglans regia L.) green husk by supercritical CO2: Process optimization using Taguchi method. J Environ Chem Eng 8(3):103776. https://doi.org/10.1016/J.JECE.2020.103776
Shrestha R, Khanal S, Poudel P, Khadayat K, Ghaju S, Bhandari A, Lekhak S, Pant ND, Sharma M, Marasini BP (2019) Extended spectrum β-lactamase producing uropathogenic Escherichia coli and the correlation of biofilm with antibiotics resistance in Nepal. Ann Clin Microbiol Antimicrob 18(1):42. https://doi.org/10.1186/s12941-019-0340-y
Silva EM, Souza JNS, Rogez H, Rees JF, Larondelle Y (2007) Antioxidant activities and polyphenolic contents of fifteen selected plant species from the Amazonian region. Food Chem 101(3):1012–1018. https://doi.org/10.1016/J.FOODCHEM.2006.02.055
Singleton VL, Orthofer R, Lamuela-Raventós RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol 299:152–178. https://doi.org/10.1016/S0076-6879(99)99017-1
Sobisch LY, Rogowski KM, Fuchs J, Schmieder W, Vaishampayan A, Oles P, Novikova N, Grohmann E (2019) Biofilm Forming Antibiotic Resistant Gram-Positive Pathogens Isolated From Surfaces on the International Space Station. Front Microbiol 10:543. https://doi.org/10.3389/fmicb.2019.00543
Zakavi F, Hagh L, Daraeighadikolaei A, Farajzadeh Sheikh A, Leilavi Shooshtari Z (2013)Antibacterial effectof Juglans regiabark against oral pathologic bacteria. Int J Dent
Zarei M, Ghanbari R, Tajabadi N, Abdul-Hamid A, Bakar F (2016) Generation, fractionation, and characterization of iron-chelating protein hydrolysate from palm kernel cake proteins. J Food Sci 81:C341–C347
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
HA and ND contributed to the study conception and design. Material preparation and analysis were performed by YO, SO, OC, and GT. YO and EOK wrote the manuscript with support from ND and SO, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arslan, H., Ondul Koc, E., Ozay, Y. et al. Antimicrobial and antioxidant activity of phenolic extracts from walnut (Juglans regia L.) green husk by using pressure-driven membrane process. J Food Sci Technol 60, 73–83 (2023). https://doi.org/10.1007/s13197-022-05588-w
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-022-05588-w